Zhang, J.; Page, A. C. S.; Palani, V.; Chen, J.; Hoye, T. R. Org. Lett. 2018, 20, 5550–5553.
Abstract
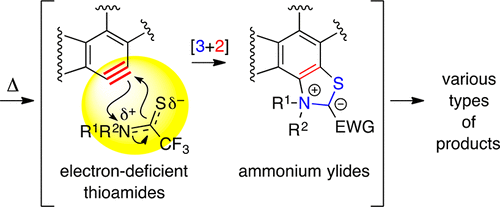
Thioamides bearing electron-withdrawing groups on the thiocarbonyl carbon atom react with benzynes [generated by the hexadehydro-Diels–Alder cycloisomerization] in an unprecedented fashion. Namely, the dihydrobenzothiazole products are consistent with a pathway involving initial formation of a stabilized ammonium ylide by a rare type of [3 + 2]-cycloaddition reaction. The fate of this species depends upon the nature of the R group(s) attached to the ylide nitrogen atom. The demonstration of new modes of reactivity represents the major advance arising from this study.