McClintock, M. K.; Fahnhorst, G. W.; Hoye, T. R.; Zhang, K. Metab. Eng. 2018, 48, 208–217.
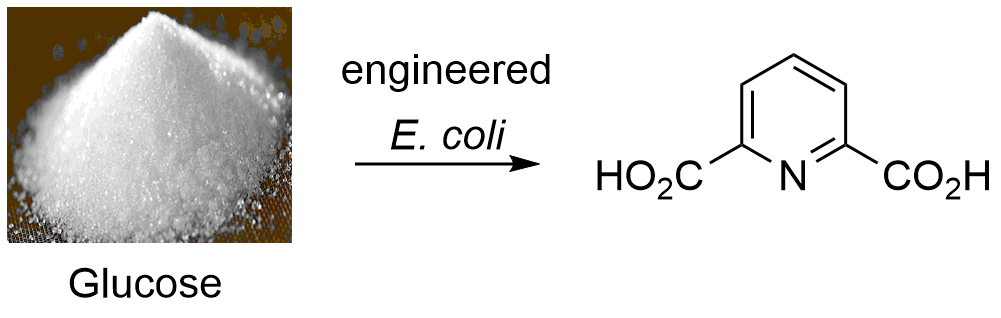
Dicarboxylic acids, such as the phthalic acids and their derivatives, are monomeric components in several important polyesters and polyamides. In most cases, these compounds are derived from fossil fuels and are not easily biodegradable. Dipicolinic acid (DPA) is a biologically derived aromatic di-acid that has a similar structure to isophthalic acid. Furthermore, DPA has been shown to make polyesters, is readily biodegradable, and is non-toxic. DPA is naturally produced by Bacillus and Clostridium species during sporulation and can comprise up to 15% of the dry weight. In this paper we demonstrate the first heterologous production of dipicolinic acid and identify the genes appropriate for gram-scale production in the industrial workhorse organism, E. coli. First, several genetic constructs were designed to identify the genes necessary for recombinant production in E. coli from the aspartate pre-cursor. The in vitro activity of dipicolinate synthase was then compared between Bacillus subtilits and Clostridium perfringens. Next, in order to improve DPA production from glucose an optimized strain was created containing the elimination of several genes resulting in 5.21 g/L DPA when 5 g/L of aspartate was supplied. Then, several aspartate kinsases and dipicolinate synthases were screened for optimal activity in E. coli. The optimal genes were combined with the overexpression of phosphoenolpyruvate carboxylase, developing a full biosynthetic pathway capable of producing a titer of 4.7 g/L DPA directly from glucose. In summary, we have performed detailed biochemical study of key pathway enzyme dipicolinate synthase and achieved scalable heterogeneous production of DPA in the workhorse organism E. coli.