Woods, B. P.; Baire, B. ; Hoye, T. R. Org. Lett. 2014, 16, 4578–4581.
Abstract
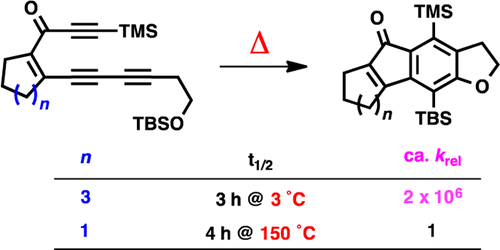
The rates of the hexadehydro-Diels–Alder (HDDA) reaction of substrates containing, minimally, a 1,3,8-triyne subunit are reported. Several series of related substrates, differing in the nature of the three-atom tether that links the 1,3-diyne and diynophile, were examined. Seemingly small changes in substrate structure result in large differences in cyclization rate, spanning more than 8 orders of magnitude. The reactivity trends revealed by these studies should prove useful in guiding substrate design and choice of reaction conditions in future applications.