Arora, S.; Zhang, J.; Pogula, V.; Hoye, T. R. Chem. Sci. 2019, 10, 9069–9076.
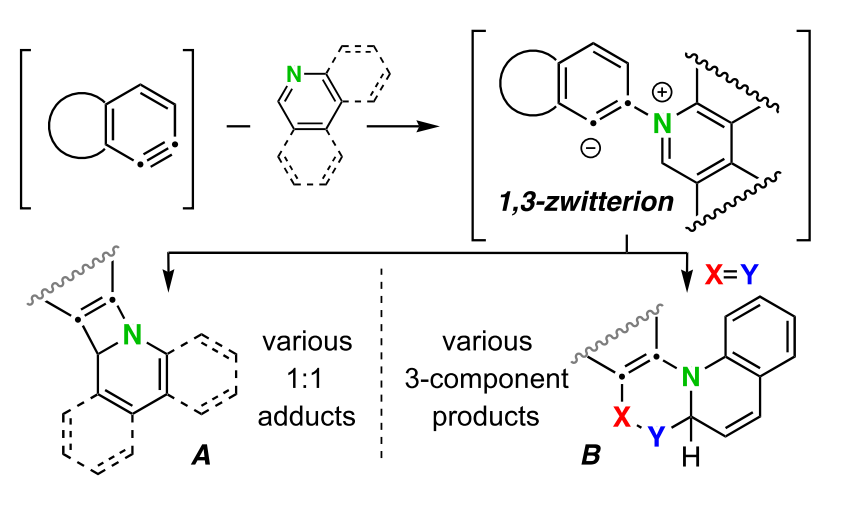
We report here various pathways by which six-membered N–heteroaromatic compounds react with benzynes that are generated by the HDDA reaction. The initially formed 1,3-zwitterionic species a) can collapse intramolecularly to give novel 1:1 adducts of the heterocycle and benzyne; b) can react with an externally added, electrophilic third-component to give functionalized heterocyclic products; or c) can react with an external protic nucleophile to produce, following collapse of the ion pair resulting from protonation of the zwitterion, a variety of three-component assemblies. Mechanisms for formation of some of the 1:1 adducts are supported by DFT methods. The scope of the protic nucleophilic coupling was also expanded to a two-pot operation by using triflic acid as a protic “non-nucleophile”, followed by the addition of a suitably reactive nucleophile.