Hassig, C.A.; Zeng, F.Y.; Kung, P.; Kiankarimi, M.; Kim, S.; Diaz, P.W.; Zhai, D.; Welsh, K.; Morshedian, S.; Su, Y.; O’Keefe, B.; Newman, D.J.; Rusman, Y.; Kaur, H.; Salomon, C.E.; Brown, S.G.; Baire, B.; Michel, A. R.; Hoye, T. R.; Francis, S.; Georg, G.I.; Walters, M.A.; Divlianska, D.B.; Roth, G.P.; Wright, A.E.; Reed, J.C. J. Biomol. Screen. 2014, 19, 1201–1211.
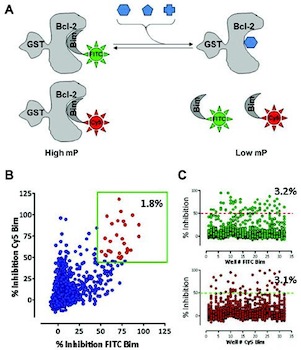
Antiapoptotic Bcl-2 family proteins are validated cancer targets composed of six related proteins. From a drug discovery perspective, these are challenging targets that exert their cellular functions through protein-protein interactions (PPIs). Although several isoform-selective inhibitors have been developed using structure-based design or high-throughput screening (HTS) of synthetic chemical libraries, no large-scale screen of natural product collections has been reported. A competitive displacement fluorescence polarization (FP) screen of nearly 150,000 natural product extracts was conducted against all six antiapoptotic Bcl-2 family proteins using fluorochrome-conjugated peptide ligands that mimic functionally relevant PPIs. The screens were conducted in 1536-well format and displayed satisfactory overall HTS statistics, with Z′-factor values ranging from 0.72 to 0.83 and a hit confirmation rate between 16% and 64%. Confirmed active extracts were orthogonally tested in a luminescent assay for caspase-3/7 activation in tumor cells. Active extracts were resupplied, and effort toward the isolation of pure active components was initiated through iterative bioassay-guided fractionation. Several previously described altertoxins were isolated from a microbial source, and the pure compounds demonstrate activity in both Bcl-2 FP and caspase cellular assays. The studies demonstrate the feasibility of ultra-high-throughput screening using natural product sources and highlight some of the challenges associated with this approach.